Gasoline, Methanol, AFR, Lambda, and Stoichiometry.
#23
Thread Starter
Joined: Sep 2005
Posts: 33,516
Total Cats: 6,913
From: Chicago. (The less-murder part.)
So, the question is:
When burning a more-or-less homogeneous mixture of gasoline and methanol, at a combined 0.85 lambda, will the gasoline behave in the same way as it would were it running all by itself at 0.85 lambda?
In other words, gasoline at 1λ (or even leaner) would be expected to produce high EGTs, detonate, etc., when run through an engine that's operating at 180 kPa. By contrast, gasoline running at 0.8λ, under the same conditions, tends to behave itself provided the ignition timing is not too aggressive. Will it continue to do so in an overall 0.85λ environment, even if the amount of gasoline present is less than would constitute a 0.85λ mixture were it not for the presence of some other fuel as well?
When burning a more-or-less homogeneous mixture of gasoline and methanol, at a combined 0.85 lambda, will the gasoline behave in the same way as it would were it running all by itself at 0.85 lambda?
In other words, gasoline at 1λ (or even leaner) would be expected to produce high EGTs, detonate, etc., when run through an engine that's operating at 180 kPa. By contrast, gasoline running at 0.8λ, under the same conditions, tends to behave itself provided the ignition timing is not too aggressive. Will it continue to do so in an overall 0.85λ environment, even if the amount of gasoline present is less than would constitute a 0.85λ mixture were it not for the presence of some other fuel as well?
#24
I think the answer to this could be close enough to the same behavior as one would see running ethanol blended fuel. So, if one fills up with say E10 or E20, uses WI (all water), and compensates for the slightly leaner condition my adjusting back to the target lambda (maybe the ECU does this on its own in some cases), is the car more knock prone, have higher EGTs, etc, or does it all more-or-less balance out?
Seems like this is a common enough scenario that an experienced tuner would know the answer. Perhaps the answer is out there in Internetland via the Google magic bus.
Seems like this is a common enough scenario that an experienced tuner would know the answer. Perhaps the answer is out there in Internetland via the Google magic bus.
#25
Thread Starter
Joined: Sep 2005
Posts: 33,516
Total Cats: 6,913
From: Chicago. (The less-murder part.)
I am finding a hell of a lot of good information on water-injection in general, much of it from NASA based on studies performed during WWII on aircraft engines. Turns out that this is, in fact, rocket science. At some point, as part of my upcoming modifications I'm planning to write a sort of thesis-style FAQ concatenating a lot of what I'm learning as well as capturing the data that I hope to generate while doing some experiments of my own. That's going to require a hell of a lot more motivation than I have just at the moment, however.
Despite all this, I have yet to find any research that specifically answers the question of how gasoline behaves in the presence of other fuels. I had not, however, considered the E10 / E85 angle, so perhaps bending my queries towards modern flex-fuel vehicles may produce some answers. The downside here is that most of the old stuff is freely available, whereas access to much of the more contemporary research is subscription-based or requires SAE membership.
That's one thing I really miss about college- having easy access to good libraries.
#26
Joined: Jun 2005
Posts: 19,338
Total Cats: 574
From: Fake Virginia
so I was browsing around in Heywood and there was no simple answer for mixed fuels.
but there are two slightly different equations for the combustion process because of the alcohol's oxygen content.
so I suppose you could write a long chemical equation using both fuels and their mole fractions.
or you could assume that the lambda is simply weighted by the fuel ratios. which I think is acceptable because you're breaking both down into their raw constituents which recombine with air and presumably aren't interacting some other way to give you your combustion products.
stoich for methanol is 6.5:1
stoich for gasoline is 14.7:1
so if you burn 90% gasoline and 10% methanol, you'd have .9(14.7) plus .1(6.5) is 13.88 as your new stoich value. and your wideband would read lambda=1 at that AFR.
but there are two slightly different equations for the combustion process because of the alcohol's oxygen content.
so I suppose you could write a long chemical equation using both fuels and their mole fractions.
or you could assume that the lambda is simply weighted by the fuel ratios. which I think is acceptable because you're breaking both down into their raw constituents which recombine with air and presumably aren't interacting some other way to give you your combustion products.
stoich for methanol is 6.5:1
stoich for gasoline is 14.7:1
so if you burn 90% gasoline and 10% methanol, you'd have .9(14.7) plus .1(6.5) is 13.88 as your new stoich value. and your wideband would read lambda=1 at that AFR.
#27
Thread Starter
Joined: Sep 2005
Posts: 33,516
Total Cats: 6,913
From: Chicago. (The less-murder part.)
I thought we all agreed that because 14.7 for gasoline is 1λ, and 6.5 for methanol is also 1λ, and because the sensor itself actually reads lambda and then the controller converts it to AFR based on a lookup table, that stoich for any homogeneous mixture of gasoline and methanol will show as 14.7 on a display gauge that has been calibrated for gasoline.
#28
Joined: Jun 2005
Posts: 19,338
Total Cats: 574
From: Fake Virginia
?!
I thought we all agreed that because 14.7 for gasoline is 1λ, and 6.5 for methanol is also 1λ, and because the sensor itself actually reads lambda and then the controller converts it to AFR based on a lookup table, that stoich for any homogeneous mixture of gasoline and methanol will show as 14.7 on a display gauge that has been calibrated for gasoline.
I thought we all agreed that because 14.7 for gasoline is 1λ, and 6.5 for methanol is also 1λ, and because the sensor itself actually reads lambda and then the controller converts it to AFR based on a lookup table, that stoich for any homogeneous mixture of gasoline and methanol will show as 14.7 on a display gauge that has been calibrated for gasoline.
sorry I've been sick and a little spaced out.
i just was hung up on finding out the specifics of the chemistry involved.
#29
I feel kind of bad for resurrecting this thread being that it is almost 5 months old. Having said that, I just have to tell you guys that reading through this conversation has not only been a pleasure, but it has also answered every single one of the questions I had sat down to research on tuning with Meth Injection. I am a Honda guy, but if you guys regularly have such intelligent and readable technical discussions (unlike the majority of my Honda-Enthusiast brethren), I will most certainly be returning to this forum.
Thanks
Thanks
#30
Elite Member
iTrader: (22)
Joined: Dec 2006
Posts: 3,224
Total Cats: 3
From: Sunny Spanish speaking Non US Caribbean
I feel kind of bad for resurrecting this thread being that it is almost 5 months old. Having said that, I just have to tell you guys that reading through this conversation has not only been a pleasure, but it has also answered every single one of the questions I had sat down to research on tuning with Meth Injection. I am a Honda guy, but if you guys regularly have such intelligent and readable technical discussions (unlike the majority of my Honda-Enthusiast brethren), I will most certainly be returning to this forum.
Thanks
Thanks

If you're looking for good info about anything mechanical (Miata related), you've come to the right place.
Go to the "Meet and Greet" section and introduce yourself properly.
Enjoy your stay.
#33
?!
I thought we all agreed that because 14.7 for gasoline is 1λ, and 6.5 for methanol is also 1λ, and because the sensor itself actually reads lambda and then the controller converts it to AFR based on a lookup table, that stoich for any homogeneous mixture of gasoline and methanol will show as 14.7 on a display gauge that has been calibrated for gasoline.
I thought we all agreed that because 14.7 for gasoline is 1λ, and 6.5 for methanol is also 1λ, and because the sensor itself actually reads lambda and then the controller converts it to AFR based on a lookup table, that stoich for any homogeneous mixture of gasoline and methanol will show as 14.7 on a display gauge that has been calibrated for gasoline.
How then would the Lambda sensor automagically adjust itself for whatever is stoich for your mixture?
#34
I made a little table showing what stoich would be for various ratios of meth to water:
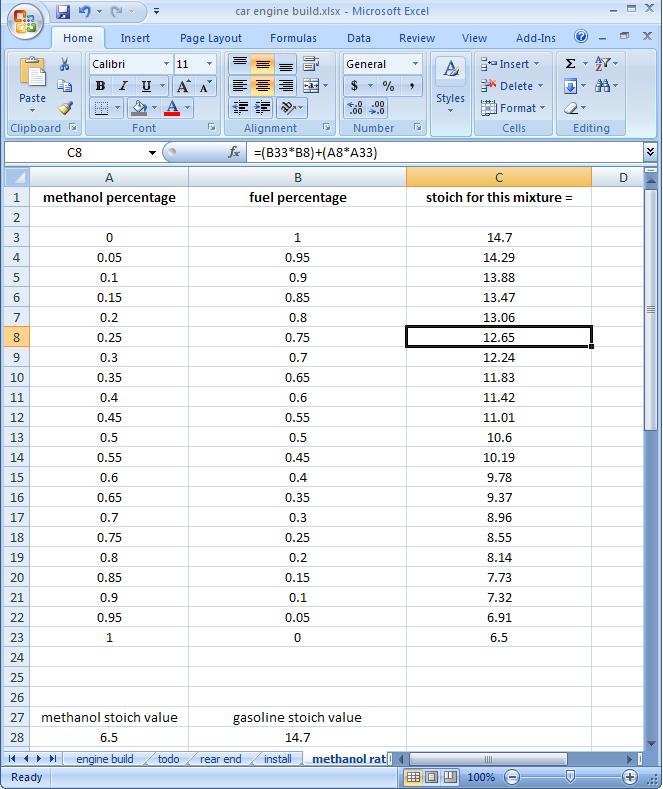
So for lambda to be properly configured for your mixture you would need to set lambda to equal the rightmost value.
Back to Joe's original question,
"Assuming our WBO2 system is calibrated for gasoline, is it safe to assume that when the display on it reads 12.5 (equating to 0.85λ) that regardless of the ratio of gasoline to methanol going into the engine, the overall ratio of the combined mixture of fuels to air is ideal?"
My answer is no. Stoich isn't necessarily doesn't provide us with best torque or safety for gas or meth.
Assuming 5.5 gives us the most power and safety for meth and 12.5 does the same for fuel, we get this table:
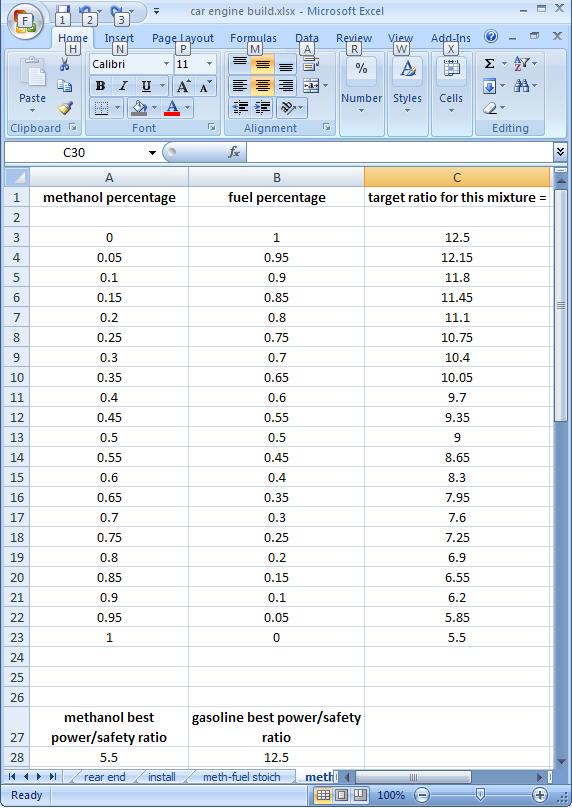
At 25% meth, 75% fuel we would be at 10.75 AFR.

So for lambda to be properly configured for your mixture you would need to set lambda to equal the rightmost value.
Back to Joe's original question,
"Assuming our WBO2 system is calibrated for gasoline, is it safe to assume that when the display on it reads 12.5 (equating to 0.85λ) that regardless of the ratio of gasoline to methanol going into the engine, the overall ratio of the combined mixture of fuels to air is ideal?"
My answer is no. Stoich isn't necessarily doesn't provide us with best torque or safety for gas or meth.
Assuming 5.5 gives us the most power and safety for meth and 12.5 does the same for fuel, we get this table:

At 25% meth, 75% fuel we would be at 10.75 AFR.
#37
Thread Starter
Joined: Sep 2005
Posts: 33,516
Total Cats: 6,913
From: Chicago. (The less-murder part.)
My quandary is this: If we assume that X represents the ideal lambda ratio for gasoline, and Y represents the ideal lambda ratio for methanol, does it follow that (X * %gas) + (Y * %meth) = the ideal lambda ratio for any given mixture of the two? Or put another way, does the presence of one fuel affect the behavior of the other insofar as ideal λ for MBT is concerned?
Thread
Thread Starter
Forum
Replies
Last Post
Motorsport-Electronics
ECUs and Tuning
0
09-05-2015 08:02 AM